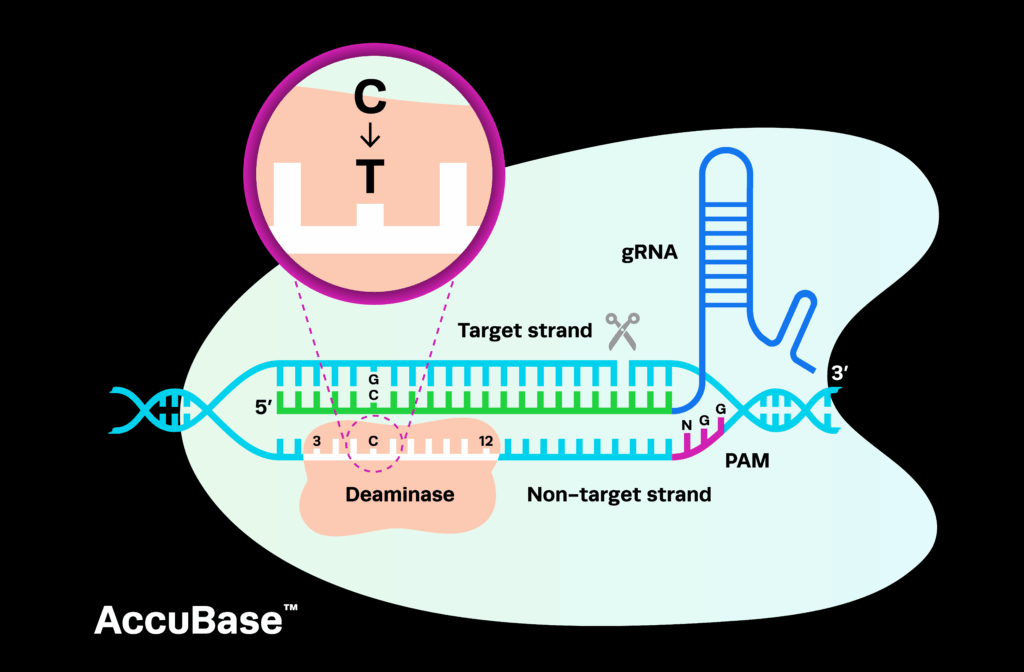
When should you choose a CRISPR delivery method? As you move from discovery to development, one decision quietly shapes everything that comes next: how you deliver your CRISPR system. Early on, it’s natural to focus on editing efficiency, specificity, and experimental success. But as programs advance, new pressures emerge, often later than expected, forcing teams to confront questions that can’t be answered by laboratory performance alone.
You may start asking:
- Can this process scale beyond a handful of experiments?
- What will this cost when applied to real patient populations?
- How complex will manufacturing become under regulatory constraints?
These questions highlight a crucial point: the delivery modality you select early on doesn’t just influence whether your CRISPR system works in the lab; it determines how scalable, cost-effective, and manufacturable your therapy can be. Many programs run into challenges not because the editing itself fails, but because the delivery strategy struggles to translate beyond early-stage success.
Delivery Modality is a Strategic Decision. Not just a Technical One
It’s easy to think of delivery as a technical variable, one of many parameters to optimize in a CRISPR workflow. In reality, it’s a strategic decision with long-term consequences.
The modality you choose influences far more than editing performance. It affects:
Manufacturing complexity: How many steps are required? How specialized is the process?
Scalability: Can production increase linearly, or does it require entirely new infrastructure?
Cost of goods (COGS): Do costs rise gradually with scale, or exponentially?
Supply chain reliability: Are you dependent on complex biological systems or modular production inputs?
Development timelines: How quickly can you move from lab-scale to clinical-grade material?
These considerations don’t just appear at later stages. They are locked in by early design choices. That’s why delivery modality should be evaluated not only for how well it works today, but for how well it will hold up as your program grows.
CRISPR Delivery Methods: Cargo, Vehicles, and Challenges
Go beyond scalability to understand the full CRISPR delivery landscape, including cargo design, delivery vehicles, and key challenges across research and therapeutic applications.
AAV: Clinically Validated, Operationally Complex
Adeno-associated virus (AAV) has been foundational in advancing gene therapy. It has enabled in vivo delivery across multiple indications and remains one of the most clinically established platforms available.
But if your goal is to scale, AAV introduces a distinct set of operational challenges.
Unlike synthetic or modular systems, AAV production depends on biological manufacturing processes. These typically include:
- Expansion of producer cells (mammalian or insect)
- Multi-plasmid transfection to introduce viral components
- Intracellular viral assembly
- Downstream purification to isolate functional particles
- Extensive quality control and release testing
Each step introduces variability, time, and resource demands. Production cycles can span weeks, and scaling often requires replicating complex infrastructure rather than simply increasing output.
What Does Scaling AAV Actually Look Like?
To understand how this plays out, consider a simplified scenario.
For certain in vivo applications, dosing requirements can be extremely high. Producing enough viral material for even a single adult patient can demand substantial bioreactor capacity. Scaling across multiple patients compounds the challenges, often requiring larger facilities, increased batch volumes, and higher risk of variability or batch failure. When capital investment, facility maintenance, and process inefficiencies are factored in, the true cost of scaling becomes difficult to predict. This doesn’t reduce the clinical importance of AAV. Instead, it highlights an important reality: AAV is powerful, but it is not inherently designed for simple or flexible scaling.
Non-Viral: Modular by Design, Built for Scale
Non-viral delivery approaches shift the paradigm. Rather than producing and purifying viral particles, CRISPR components are generated using defined, biochemical processes, such as:
- Recombinant protein expression for nucleases
- Enzymatic in vitro transcription for mRNA
- Synthetic production of guide RNAs
These components are then combined with delivery systems like lipid nanoparticles (LNPs). The difference isn’t just technical; it’s structural. Non-viral systems are built on modular manufacturing principles.
This means:
- Production steps are more standardized
- Scaling often involves increasing batch size, not redesigning infrastructure
- Processes can be more predictable and repeatable
While non-viral approaches are still evolving, they offer a pathway that aligns more naturally with modern, scalable biomanufacturing frameworks. For teams thinking ahead to clinical translation, this shift can reduce friction between early discovery and later-stage production.
Protein and mRNA: Complementary Paths within Non-Viral Delivery
Within non-viral CRISPR delivery, protein and mRNA represent two distinct but complementary strategies. While both enable effective gene editing, they differ in how the editing machinery is introduced and controlled; making it important to align the approach with your program’s specific priorities.
Ribonucleoprotein (RNP) delivery introduces the CRISPR nuclease directly as a pre-formed protein complex. Because the editing machinery is delivered in its active form, editing can begin almost immediately after delivery, with tight control over how long the nuclease remains active in the cell. This approach minimizes reliance on cellular expression mechanisms, which can reduce variability and improve predictability at the point of editing. As a result, RNP has become a well-established standard across many research and translational workflows where precision and control are critical.
In contrast, mRNA delivery leverages the cell’s own machinery to produce the nuclease internally. Rather than delivering the protein itself, mRNA is introduced and translated within the cell, enabling a more transient and tunable expression profile. Because mRNA is generated through enzymatic synthesis, it integrates seamlessly with nucleic acid manufacturing platforms and can be scaled using well-established processes. This makes mRNA a flexible option for applications where controlled expression and streamlined production are key considerations.
Advance Your CRISPR Strategy with Protein and mRNA
Partner with Synthego to access high-quality CRISPR components designed to support flexible, scalable delivery across protein and mRNA workflows.
How Do You Choose the Right Delivery Method?
Selecting the right CRISPR delivery method isn’t just a technical decision; it’s a strategic one. The optimal approach depends on what success looks like for your program and what it will take to get there. Early discovery, process development, and clinical manufacturing each introduce different constraints, and your delivery strategy needs to support that full progression.
Viral vectors such as AAV have long been a foundation in gene editing, offering efficient in vivo delivery and a well-established clinical track record. For certain applications, particularly where long-term expression is required, AAV remains a powerful and proven option.
At the same time, non-viral CRISPR delivery methods, including protein and mRNA, are expanding what’s possible, particularly in contexts where control, flexibility, and scalability are critical. Protein delivery enables immediate editing activity and tight temporal control, making it well-suited for applications where precision and predictability are essential. In contrast, mRNA delivery leverages cellular machinery to produce the nuclease internally, offering a more flexible and scalable approach that integrates naturally with nucleic acid manufacturing workflows.
Rather than a one-size-fits-all solution, these modalities collectively form a broader toolkit. As you evaluate your options, it’s important to ask: What does success look like for this program? What will it require to get there? Increasingly, the ability to choose and adapt your delivery strategy over time is not just helpful, it’s becoming a competitive advantage.
How Synthego Supports Delivery Decisions
Choosing between CRISPR delivery methods is rarely a one-time decision. As programs advance, requirements shift from optimizing early-stage performance to meeting the demands of process development and clinical manufacturing. Having the flexibility to evaluate and adapt your approach along the way can reduce risk and improve long-term outcomes.
This flexibility is also becoming a clear competitive advantage. Teams that can move between modalities, optimize at each stage, and avoid early constraints are better positioned to scale efficiently and respond to evolving program needs.
Synthego supports this approach by providing high-quality CRISPR components across both protein and mRNA formats. This enables teams to explore and optimize different delivery strategies while maintaining consistent reagent quality and performance. Whether you’re prioritizing the precision and control of RNP or the scalability and integration advantages of mRNA, you can align your approach to the needs of each stage of development.
By working with a partner that supports multiple modalities, you’re better positioned to avoid being locked into a single pathway too early and instead build a strategy that evolves with your program.
Design for Scale from the Start
The future of gene editing will not be defined by a single delivery technology; it will be shaped by how effectively scientific innovation translates into scalable, manufacturable therapies.
As you evaluate CRISPR delivery methods, it’s worth stepping back and asking: Will this approach scale beyond early success? Can it support clinical and commercial demand? Does it align with long-term manufacturing realities?
Because in the end, the delivery method you choose today will shape your ability to deliver therapies tomorrow.