You’ve probably heard that CRISPR is transforming gene editing—but it’s also starting to change how diagnostics are built and used.
If you’re working in diagnostics, you’re likely balancing familiar tradeoffs: speed versus sensitivity, accessibility versus precision, and performance versus scalability. Traditional methods like PCR and antigen testing are still highly effective, but they often force you to prioritize one over the other. CRISPR offers a different approach. Instead of replacing existing methods, it gives you another tool—one that’s programmable, adaptable, and easier to deploy in a wider range of settings. In this guide, we’ll walk through how CRISPR diagnostics work, where they add the most value, and how you can apply them within your own workflow.
How Do CRISPR-Based Diagnostics Work?
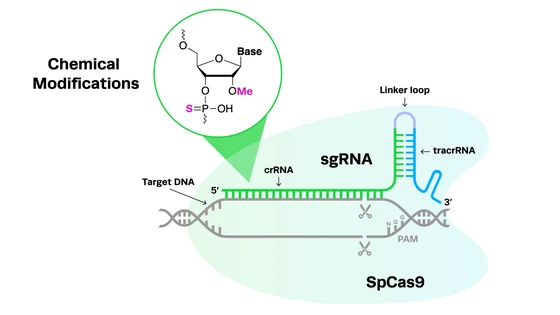
At a high level, CRISPR diagnostics give you a programmable way to detect specific genetic targets. You design a guide RNA (gRNA) to match the sequence you care about, and CRISPR-associated enzymes—typically Cas12 or Cas13—use that guide to find it in your sample. In most workflows, this detection step is paired with an amplification stage, such as RPA or RT-RPA, to increase the amount of target material and improve sensitivity. Once the target is present in sufficient quantity, the CRISPR system activates and produces a detectable signal—either through fluorescence or a visual readout like a lateral flow strip.
The result is a fast, specific way to determine whether your target is present—without the need for complex thermal cycling or heavy lab infrastructure.
That’s the high-level view. If you want a deeper dive into how each step works—from amplification through signal detection—check out our full technical guide: How to Build an RPA-CRISPR Assay for Rapid Diagnostics.
How Does CRISPR Compare to Traditional Diagnostic Methods?
If you’re developing a diagnostic, you’ve probably run into the same challenge: balancing sensitivity, specificity, speed, and accessibility. Traditional methods tend to force tradeoffs—PCR offers strong sensitivity and quantification but requires more infrastructure, while rapid tests are faster and simpler but can miss low-abundance targets. CRISPR gives you more flexibility in how you approach that balance. When paired with amplification methods like RPA or RT-RPA, it allows you to achieve strong sensitivity without the complexity of PCR, while keeping workflows relatively fast and flexible. And because CRISPR is programmable, you can adapt your assay more quickly as targets change—without starting from scratch.
So rather than choosing one approach over another, the real question becomes: where does each method fit best in your workflow?
Where CRISPR Diagnostics Add the Most Value
CRISPR-based diagnostics really start to stand out when speed, flexibility, and accessibility matter. If you’re working in a setting where timelines are tight, targets are evolving, or testing needs to happen outside of a traditional lab, CRISPR gives you more room to adapt. Instead of forcing you into a single workflow, it lets you design based on your specific needs, whether that’s faster turnaround, simpler deployment, or the ability to pivot quickly as things change.
- Get fast results without giving up specificity: By pairing RPA or RT-RPA with CRISPR detection, you can generate results in under an hour while still targeting highly specific genetic sequences.
- Design assays around exactly what you need to detect: The gRNA acts as a programmable component, giving you precise control over which pathogens, variants, or mutations your assay identifies.
- Adapt quickly as targets evolve: Instead of rebuilding your entire system, you can often update your assay by redesigning the gRNA—making it much easier to respond to emerging threats.
- Move testing beyond the lab: Because CRISPR-based assays don’t require complex thermal cycling, they can be used in point-of-care, field, or resource-limited environments.
- Detect multiple targets in a single workflow: With the right design, you can build multiplexed assays that give you more information from a single sample with a single test.
Best-in-Class CRISPR Solutions
Explore Synthego’s portfolio of synthetic guide RNAs, and bring unmatched reproducibility and performance to your development pipeline.
What You Should Keep in Mind
At the same time, it’s important to be aware of where CRISPR requires careful optimization. One of the main challenges is the potential for off-target effects. In some cases, gRNAs can tolerate small mismatches, which may lead to unintended signal generation if the assay isn’t fully optimized. For you, this means that performance depends heavily on how well your gRNA is designed and validated. Investing upfront in guide selection and validation can significantly reduce variability and improve reliability, especially as you move toward real-world use.
Where Traditional Methods Still Excel
CRISPR brings a lot of flexibility, but traditional diagnostic methods still play an important role in many workflows. In many cases, they’re still the right tool for the job.
- Exceptional sensitivity for low-abundance targets: When you need to detect very small amounts of genetic material, PCR is still the gold standard.
- Reliable quantification of viral or bacterial load: Techniques like qPCR don’t just tell you if something is present—they tell you how much is there, which is critical for many clinical and research applications.
- A clear path through regulatory and clinical workflows: With decades of validation and widespread adoption, PCR is often easier to integrate into regulated environments.
- Familiar, standardized processes across labs: Most labs are already set up to run PCR, which can simplify implementation and scaling.
CRISPR vs. Traditional Diagnostics at a Glance
|
Feature
|
CRISPR (with RPA)
|
PCR
|
Rapid Antigen Tests
|
|
Target
|
DNA/RNA
|
DNA/RNA
|
Protein
|
|
Speed
|
Fast (<1 hour) |
Moderate (hours)
|
Very fast (minutes)
|
|
Sensitivity
|
High (with amplification)
|
Very high
|
Moderate
|
|
Quantification
|
Limited
|
Strong
|
None
|
|
Equipment Needs
|
Low to moderate
|
High
|
Minimal
|
|
Portability
|
High
|
Low
|
High
|
|
Adaptability
|
Very high (programmable gRNA)
|
Moderate
|
Low
|
Looking for RPA Kits for your CRISPR Diagnostic?
Synthego offers a portfolio of customizable RPA kits engineered for maximum efficiency and specificity. Our tools are designed to streamline and adapt to your workflow to deliver the quality your application demands.
Key Considerations When Establishing a CRISPR Diagnostic
So what actually goes into building a CRISPR-based diagnostic—and how do you make sure it performs reliably beyond the lab?
It starts with a few core decisions. One of the first is choosing the right CRISPR enzyme. In most cases, Cas12 is used for DNA targets, while Cas13 is better suited for RNA, including many viruses. From there, gRNA design becomes critical. Because the gRNA determines what your assay detects, even small mismatches can impact specificity, so careful design, testing, and validation are essential.
You’ll also need to think about how results are going to be read. If you’re working in a lab setting, fluorescence-based detection may make the most sense. But if your goal is point-of-care or field deployment, lateral flow formats offer a simpler, more accessible option. At the same time, real-world samples introduce their own challenges. Blood, saliva, or environmental samples can contain inhibitors or variability that affect performance, so making sure your assay works consistently across sample types is key.
As you move from early development to real-world use, the focus shifts from “does it work?” to “does it work every time?” Scaling a CRISPR diagnostic means thinking about reproducibility, manufacturability, and consistency across batches. Stability also becomes increasingly important, especially if your assay needs to be stored, transported, or used outside of controlled lab environments, where lyophilized (lyo) components can help maintain performance and stability. Variability, especially in components like gRNA, can directly impact results, which makes quality control and standardized processes increasingly important. You’ll also need to consider validation and regulatory requirements, depending on where and how your diagnostic will be used.
Planning for these factors early can save significant time later, helping you build a diagnostic that not only works in development, but holds up in real-world conditions.
How Can Synthego Accelerate Your Diagnostic Workflow?
Developing a CRISPR-based diagnostic requires more than a strong concept—it depends on how reliably each component performs within your workflow. From amplification through detection, consistency and reproducibility are critical to achieving results you can trust at scale. Synthego supports your development process by providing integrated solutions across both target amplification and CRISPR detection, helping you reduce variability so your assay performs more consistently from development through deployment.
Why choose Synthego?
- Integrated, lyophilized amplification and detection solutions: Synthego’s lyophilized RPA and RT-RPA kits enable rapid, isothermal amplification of DNA and RNA targets, improving sensitivity upstream of CRISPR detection while simplifying storage, transport, and workflow integration.
- High-quality synthetic guide RNAs: Designed for precision and consistency, Synthego’s gRNAs support reliable target recognition and reduce variability across experiments and batches.
- Scalable manufacturing from research to deployment: Whether you’re optimizing an assay or preparing for larger-scale production, Synthego’s synthesis platforms support reproducibility at every stage.
- Custom optimization aligned to your workflow: Synthego offers tailored support across both gRNA design and RPA/RT-RPA kit configuration, helping you optimize performance based on your target, sample type, and deployment environment.
- Expert support across your workflow: From early assay design through validation and scale-up, Synthego provides guidance to help you troubleshoot and refine your system efficiently.
By providing reagents and customizable solutions for both amplification (RPA/RT-RPA) and CRISPR detection (gRNA & nucleases), Synthego helps you build a more cohesive and reliable workflow—so you can focus on performance, validation, and scaling rather than managing variability across components.
Customize your RPA Kit
Fully customized RPA solutions—from formulation and mastermix optimization to custom filling. Designed for your application; ready for your workflow.
Where Are CRISPR Diagnostics Making a Real-World Impact?
Two platforms helped put CRISPR diagnostics on the map. SHERLOCK, which uses the Cas13 enzyme, was initially developed and validated in multiple applications including the detection of Zika and Dengue viruses. The technology was later refined to include simple paper-strip readouts, and in 2020, it was deployed via an FDA Emergency Use Authorization (EUA) to detect SARS-CoV-2. DETECTR, built on Cas12, has been used to identify different strains of HPV, helping distinguish between high-risk and low-risk types in under an hour. Both examples show how CRISPR can deliver molecular-level detection without the heavy infrastructure typically required for PCR.
Beyond these early demonstrations, CRISPR-based diagnostics are now being explored across a growing range of real-world applications. These include:
- Pandemic response: enabling fast, on-site detection of emerging viral threats
- Oncology: helping identify genetic mutations linked to cancer earlier
- Food safety: detecting contamination directly in production environments
- Environmental monitoring: tracking harmful microbes in water and other samples
What connects all of these use cases is the need for faster, more adaptable testing—often outside of traditional lab settings. That’s where CRISPR continues to stand out, helping bridge the gap between what’s possible in the lab and what’s needed in the real world.
What's Next for CRISPR Diagnostics?
CRISPR diagnostics are evolving quickly, and that momentum is opening up new possibilities for how and where testing can happen. Advances in enzyme design are improving sensitivity and specificity, while new formats are making it easier to deploy tests outside of traditional lab settings—from point-of-care to at-home use. At the same time, better multiplexing and integration with digital tools are helping you get more information from each sample and make faster, more informed decisions.
For you, this means more flexibility in how you design and scale your diagnostic workflow. As CRISPR continues to mature, having reliable, consistent components—like high-quality gRNA and robust amplification—becomes even more important. Synthego’s synthetic gRNAs and customizable molecular reagents are built to support that consistency, so you can focus on developing diagnostics that are fast, adaptable, and ready for real-world use at scale.