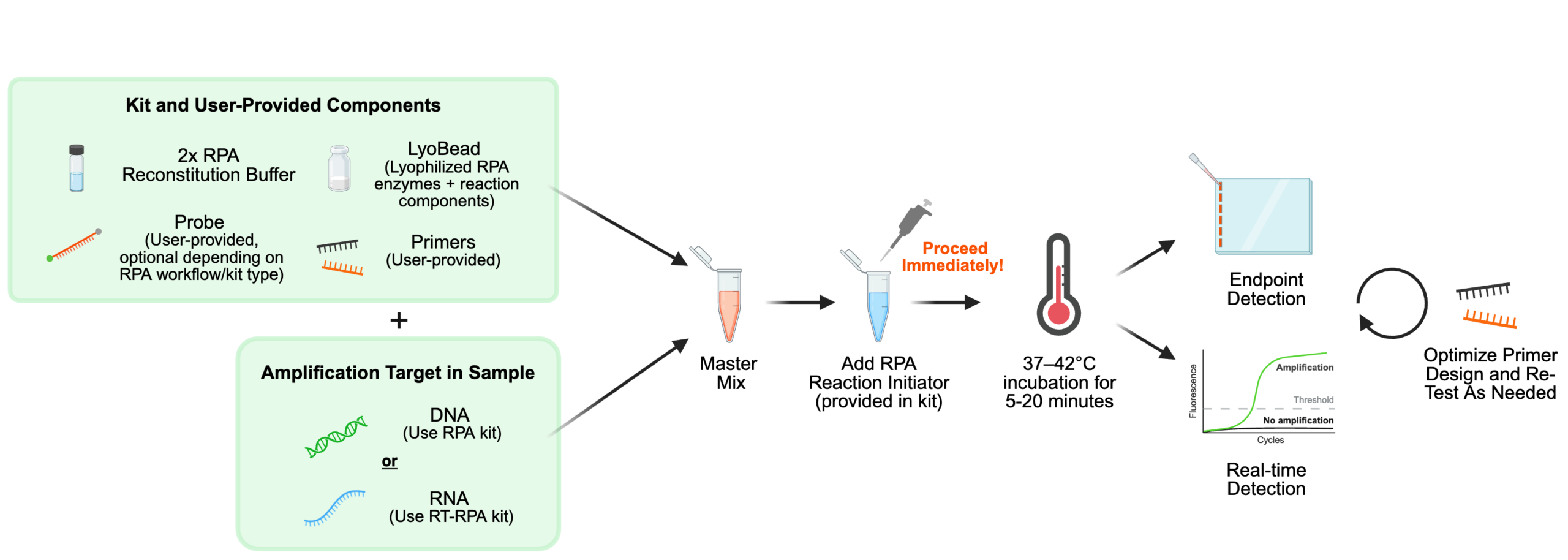
Endpoint RPA supports several detection strategies, allowing you to tailor the readout.
- Lateral Flow Assays (LFA): A gold standard for rapid, point-of-care diagnostics, lateral flow assays are most commonly used with RPA through dual-labeled amplicons that enable simple, visual detection on a test strip. This approach requires minimal equipment and delivers fast, easy-to-interpret results, making it ideal for field and low-resource settings.
Note: An emerging enhancement to this workflow leverages RPA–CRISPR synergy. In this approach, the amplified product activates a CRISPR-associated enzyme (e.g., Cas12a), which cleaves a reporter molecule via collateral (“trans”) cleavage activity. This added detection layer can improve sequence specificity and reduce background signal from non-specific amplification or primer-dimer artifacts.
- Colorimetric Detection: This method relies on a visual color change to indicate successful target amplification. By incorporating specific dyes or gold nanoparticles, you can instantly verify the presence of a target sequence.
- Electrophoresis: For laboratory settings, gel electrophoresis provides a reliable way to visualize products and confirm exact fragment sizes during the initial assay development and optimization phases.